Within the life sciences industry, controlling biological and micro contaminants in process streams is vital to improving operating efficiency and the enablement of technological advances such as modern drug development and monoclonal antibodies (mAbs). Sterile filtration plays a critical role in protecting vital bioprocesses and is the cornerstone in eliminating contaminants. But what happens when a filter gets plugged and how can you minimize impact on filtration performance?
Fundamentally, sterile filtration is the process of removing microorganisms from a fluid stream without adversely affecting your biological product. Process sterile filters use a sterilizing grade membrane with a 0.2 micron retention rating to remove biological contaminants through different retention mechanisms. These filter retention mechanisms include contaminant size exclusion and adsorption. The challenge with filter plugging is finding an optimal filtration combination that can maximize filtration life span without impacting product quality or interrupting a manufacturing process with filter change outs. Essentially, after flowing through the filter all contaminants are retained in the filter and the clean, new product streams out.
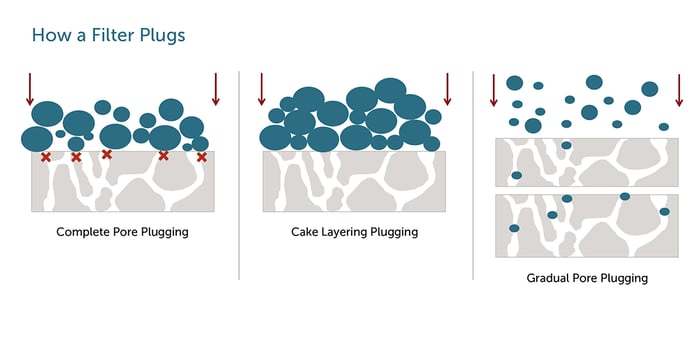
So, why do filters become plugged or blocked during intended application use? Contaminants in a product stream have particle size distribution that varies from large to small when comparing to a pore rating of a filter. Some distributions are either wide, narrow, or even and they have an impact on how plugging occurs on the filter membrane. The three most common sources of filtration failures include:
- Complete pore plugging - Pore plugging occurs when the accumulated particulates are larger than the membrane pore size and completely block the pore.
- Cake layer creation - Once complete pore plugging has occurred, particles start to stack and create layers on top of the membrane, further plugging and blocking the flow through filter.
- Gradual pore plugging - This occurs when smaller and larger particles accumulate throughout and on the surface of the membranes pore structure, gradually plugging the filter.
System and filtration parameters affect how a filter will function. Some of these parameters include system pressure and flow, fluid characteristics, membrane pore morphology, etc. Filters are designed to deliver superior flow rates in a broad range of conditions and their failure can increase operational costs, reduce yield, cause regulatory compliance concerns, and increase the cost of ownership. While all filters are designed to remove contaminants, not all filters tackle complex application challenges equally.
To prevent pore plugging or cake layer, start with an expert in contamination control technologies across a wide range of industries and applications to identify the unique solution to optimize your process. Filters are designed for a finite lifetime and understanding how to select the appropriate retention level, pre-filters, and sizing your filtration process will minimize the amount of plugging observed in your processing step. Selecting the right filter type and size ensures the appropriate filtration life span and/or performance and prevents premature failures within your processing step. Plugging for biological contaminants can occur on the surface and throughout the membrane pores and typically follows a gradual pore plugging model.
To minimize plugging and maximize filtration performance, Entegris has technical expertise and application testing capabilities to help you select the correct filter for your biological process stream. Leverage this expertise gained from decades of meeting the demanding requirements of the semiconductor industry and our dedicated life sciences business to help you fit your process with our cartridge and capsule filters. Manufactured to maximize filtration performance with high quality process controls, our filters address contamination control needs within bioprocessing, cell and gene therapy, and pharmaceutical applications.
Learn how our cartridge and capsule filters excel at filtering many common biological solutions.




